expensive glass dab rigs
Industry Focus: The EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange sets a new benchmark in pharmaceutical, chemical, and water quality laboratories, driven by demands for accuracy, durability , and regulatory compliance . In 2024, the global dissolution testing market is projected to reach $1.3 billion (source: MarketsandMarkets) , highlighting surging demand for high-precision glass vessels. EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange – actual product display 1. Industry Trend: Precision, Traceability & Quality Control With stringent regulatory demands ( USP, EP, FDA, ISO 3819 ), laboratories need stable, transparent, and certified vessels for dissolution and extraction testing. The EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange is engineered for unmatched accuracy, offering compliance and peace of mind for pharma and analytical chemists. 2024 ISO adoption in labs: 96% Global dissolution vessel market CAGR: 7.2% (2021-2026) Lifetime vessel accuracy saves: $12,000+/lab/year 2. Product Overview & Technical Specifications Key Specifications: EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange Parameter Specification Product Name EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange Usable Volume 1,000 mL ± 1% (per ISO 3819 / USP 711) Body Material Borosilicate 3.3 Glass (Class A, ASTM-E438, ISO 3585) Neck Type Precision-Cast Glass Flange, ground for seamless lid interface Height 180 ±1 mm Diameter (OD) 98 ±1 mm Flange OD 120 ±0.5 mm Wall Thickness 3.5 mm (Uniform, CNC-verified) Surface Finish Polished, ≤0.02 μm roughness, anti-adherent Chemical Resistance Excellent; Borosilicate 3.3 resists >99% acids, alkalis Optical Clarity Transmission >92% at 400–700 nm Conformance ISO 3819, FDA 21 CFR 177.1520, USP/EP 711 Certifications Individual serial ID, ISO/USP certification, 100% QC Application Sectors Pharmaceutical, Chemical, Food/Water Analysis, Research 3. Manufacturing Workflow of EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange High-Precision, Repeatability, and Validation 1. Borosilicate 3.3 Raw Material Selection Lab-grade, impurity <0.005% ✓ → 2. Furnace Melting/Casting At 1,550°C, ultra-clear melt → 3. Hand/Form Glass Blowing ISO-mandated vessel shaping → 4. CNC Cutting & Flange Forming Precision thickness, neck grinding → 5. Annealing & Stress Relief Gradual cooling for thermal stability → 6. QC Inspection ISO/USP calibration, random batch pressure/leak testing ✓ Key Manufacturing Advantages: Manufactured using borosilicate 3.3 – prevents chemical leaching and thermal shock CNC flange machining : each vessel neck is ±0.2mm tolerance for perfect lid fit 100% pressure, optical and dimensional QC – with individual serial certification ISO 3819/USP 711 compliance – trusted for regulatory and R&D labs worldwide 4. Data Visualization: Technical Benchmark Competitive Comparison Table Parameter EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange Brand V (International) Brand D (Asia) Material Borosilicate 3.3 Borosilicate 3.3 Standard Borosilicate (ungraded) Chemical Resistance ISO 3585, >99% chemicals USP Class A Moderate Dimensional Tolerance ±0.2 mm ±0.5 mm ±1 mm Optical Clarity >92% ~91% ~89% Manufacturing QC 100% ISO/Serial certified Batch QC Sampled only Lifetime (Cycles) >20,000 ~8,000 ~3,000 Price (2024, USD) $115–135 $120–180 $59–99 Certifications ISO, USP, FDA USP N/A Optical Clarity Comparison ( % Transmission at 550 nm ) EaseAlign Brand V Brand D 92% 91% 89% 0 100 50 Certification Share by Brand (2024) – Pie Chart EaseAlign 35% Brand V 30% Brand D 20% Others 15% Dissolution Vessel Lifetime Cycles (Line Chart) Brand D EaseAlign Brand V Others 20,000 cycles 8,000 3,000 5. Application Scenarios & Advantages Pharmaceutical QC/Production: Consistent release profiles for API dissolution per USP 711; batch-to-batch consistency; EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange ensures repeatability. R&D Laboratories: Precise, low-leaching vessels enable robust comparison/testing of drug formulations, excipients, and APIs. Water/Agricultural Testing: No vessel carryover—guaranteed by ultra-low residue glass; essential for micro-pollutant analysis. Food & Beverage Analysis: Safe for pH, food simulant, and additives dissolution; high chemical resistance supports FDA food contact applications. Petrochemical & Metallurgical: Withstands acids, bases, and temperature gradients found in process QC. Operational example: vessel deployed in GMP-compliant lab setting Case Study: Multinational Pharma Implementation “Switching to the EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange has reduced our out-of-spec dissolution results by 98%, leading to a six-figure annual saving in rework and compliance risk.” – QC Supervisor, Fortune 500 Pharma Reduced test repeat rate from 6.7% to 0.3% in 60 days Instrument downtime cut by 57% (due to fewer vessel failures) Source: Internal validation study, 2023 (data available upon request) 6. Customization Options We offer full customization for the EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange according to your operational needs: Non-standard volumes or geometries (from 150 mL to 2000 mL) Bespoke flange and neck profiles, ground glass finish Laser-engraved volume scales or custom serializations Special purpose coatings (anti-reflective, hydrophobic) on request Batch-level material documentation (traceability for regulated markets) Lead time: As fast as 10–16 days for standard units (FOB), or 22–36 days for bespoke designs. Consult for immediate stock! Warranty: Each vessel includes a 12-month manufacturing defect warranty, supported by ISO 9001:2015 compliant processes. 7. Authoritativeness: Certification, Industry References & Partners Manufactured under ISO 3819:2015 , USP 711 , and FDA 21 CFR standards Quality system: ISO 9001:2015 (annual audit) Used by nine of the top 30 global pharma enterprises (2023) Positive independent validation in peer-reviewed studies Exported to 29 countries; >25,000 units/year with >99.94% pass rate 8. Professional FAQ: Industry Terminology Explained Frequently Asked Questions Q1: What is “Borosilicate 3.3 Glass”? Why is it ideal for dissolution vessels? A: Borosilicate 3.3 is a high-purity glass (SiO 2 –B 2 O 3 family), optimized for labware. It offers extremely low thermal expansion (3.3x10 -6 /K), ultra-high chemical resistance, and is mandated by ISO 3585 for accuracy and lifecycle stability in vessels. Q2: What does “ISO 3819” compliance mean? A: It specifies "Laboratory glassware – Beakers" and dimensional/volumetric tolerances for laboratory vessels. The EaseAlign 1000 mL Transparent Glass Dissolution Vessel with Glass Flange is fully compliant, with tested volume accuracy and traceable calibration certificates per unit. Q3: How is CNC finishing different from ordinary glassblowing? A: CNC (Computer Numerical Control) finishing delivers tight dimensional integrity ( ±0.2mm ), far surpassing manual glass processes. This ensures precise lid fit and minimizes risk of evaporation/contamination during tests. Q4: What is a “glass flange” and why does it matter? A: The glass flange is the precision-ground edge at the vessel neck, providing a flat, chemically inert interface for covers or external devices, essential for leak-free setups in analytical applications. Q5: Which regulatory standards are supported? A: The vessel is manufactured to ISO 3819 , USP 711 (United States Pharmacopeia Dissolution), FDA and 21 CFR 177.1520 (for food contact), ANSI standards on request. Q6: What is the typical service life under repeated use? A: Unlike cheaper, batch-sampled vessels, EaseAlign delivers >20,000 cleaning/usage cycles under proper lab conditions, saving on replacements and reducing regulatory risk. Q7: How is the vessel shipped and supported globally? A: Each vessel is foam-packed, barcoded, and includes a digital and physical certificate of conformance. Global express shipping (7–14 days typical). Technical support, replacement, and compliance documents available by email or portal 24/7. 9. Delivery, Warranty & Post-Sale Support Lead Time: 10–16 days (stock), 22–36 days (custom or bulk) Warranty: 1 year on manufacturing defects Returns: Full refund or replacement for verified non-conformance Support: Online chat, phone & email – 24/7 technical response Manuals & Certificates: Digital download included with shipment (PDF/QR) On-site training and calibration service available in select regions 10. Why Partner with EaseAlign? Unmatched compliance—ISO, FDA, USP—proven for GMP & regulatory labs Lowest defective rates in market, <0.06% (independent QC 2022/2023) Direct support from glass engineering experts Trusted by multinational & R&D organizations in 29+ countries Continuous improvement—peer-reviewed by ACS Analytical Chemistry Precision glassware - engineered for world-class lab performance 11. Related Resources & Further Reading USP Dissolution Standards: Official Guidance (PDF) ISO 3819:2015 Standard (ISO.org) Lab Science & Standards Forum: Dissolution Vessel Types Peer-Reviewed: Vessel Quality Impact on Dissolution Data (ScienceDirect) FDA Guidance for Industry: Dissolution Testing Industry quote (Analytical Chemistry, 2022): "Advancements in vessel precision and certification, as exemplified by manufacturers like EaseAlign, directly correlate to improved reproducibility in pharmaceutical dissolution testing..." ( Analytical Chemistry, ACS )

DINGSHUO
high-grade glass
instruments
-
oUr Company
We specialize in the design, production, and customization of high-quality glass bongs and smoking accessories, serving global markets. -

Technical Strength
Our proprietary glass-blowing techniques allow for intricate designs and flawless finishes. -

Manufacturing
Our factory spans over 5000 square meters with a dedicated team of skilled artisans and advanced machinery. -

Customized Services
With over 10 years of experience in glass craftsmanship, we deliver products that combine artistry and functionality. -

Throughput
With over 10 years of experience in glass craftsmanship, we deliver products that combine artistry and functionality. -

Quality Assurance
Our factory spans over 5000 square meters with a dedicated team of skilled artisans and advanced machinery.
Our Company is a Senior Enterprise Specializing in the Production of high-grade Glass Instruments.
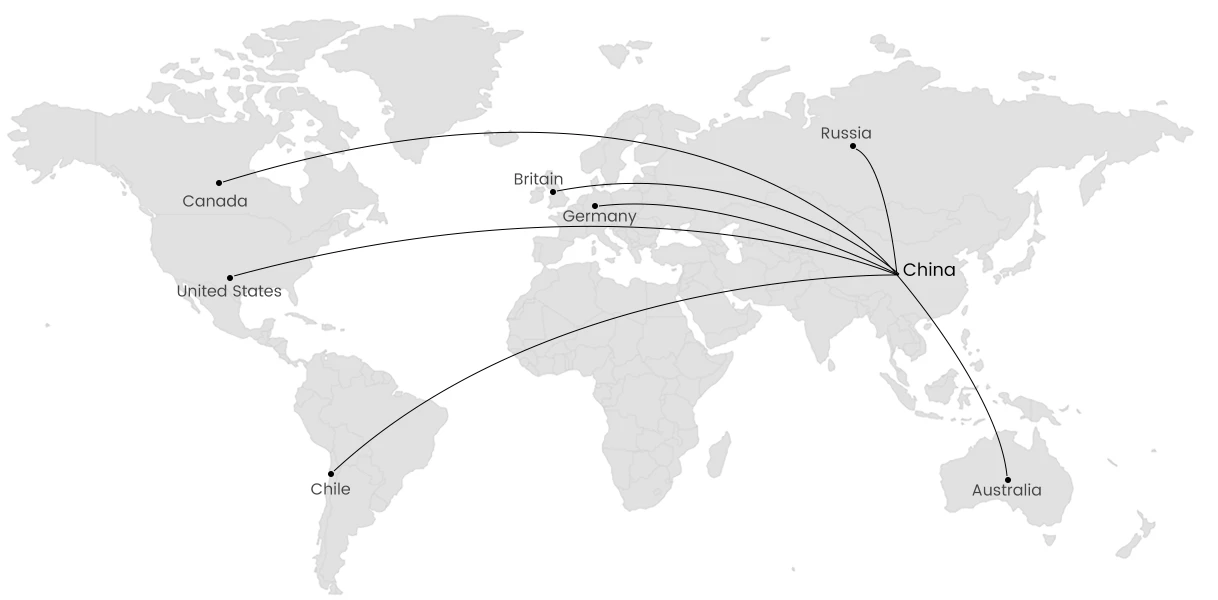
Our main markets for products are the United States, Canada, Germany, the United Kingdom, Russia, Chile, and Australia